 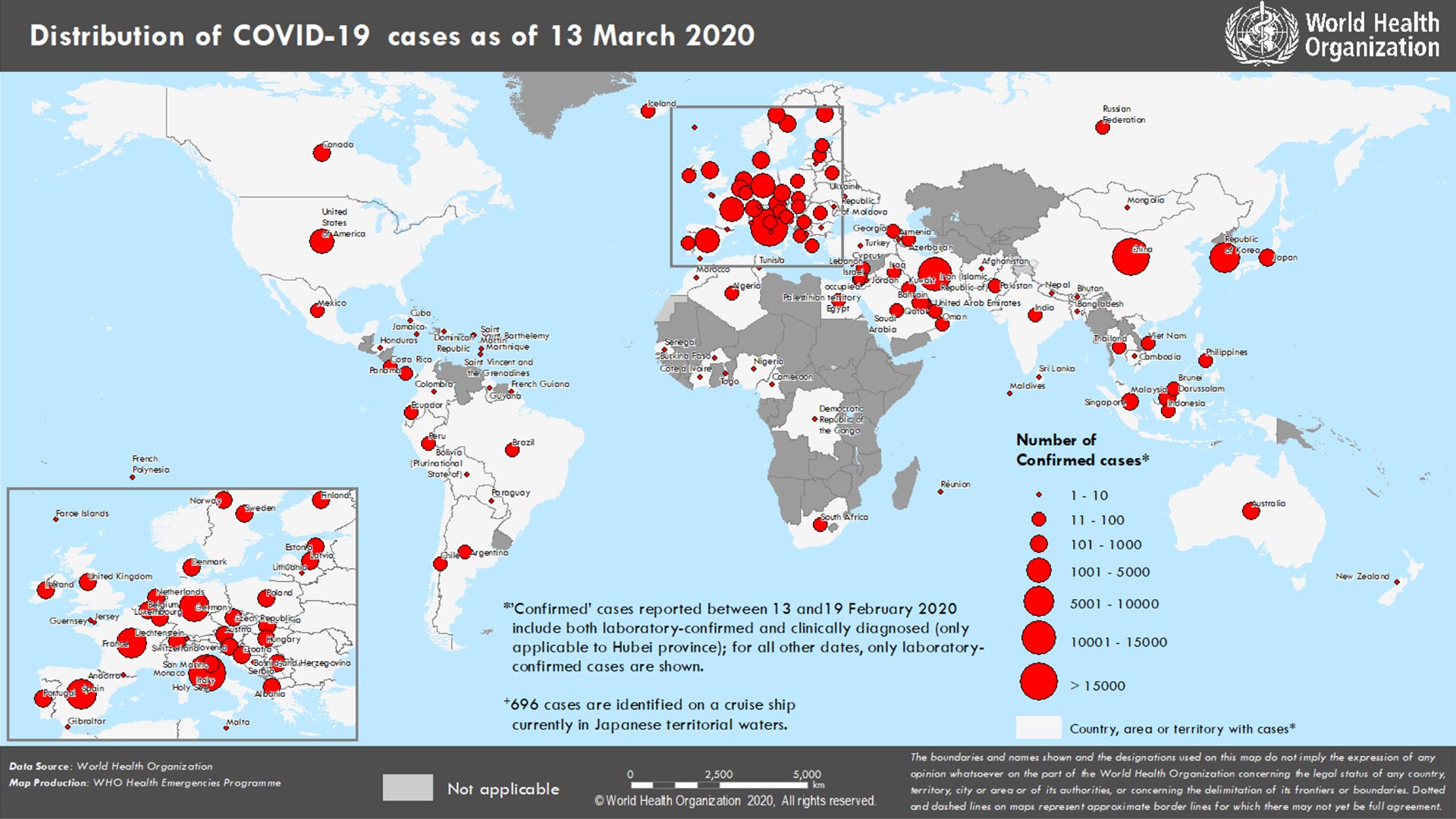
Lack of match-up was among the reasons some deaths were not included in the data published by FDOH. “As subsequently described, the number of entities reporting data, apparent inaccurate or incomplete data reported to the State by those entities, and the lack of effective access controls in the systems used to gather data, impacted the State’s ability to accurately report COVID-19 data at the beginning of the pandemic.” “To evaluate the State’s readiness to provide essential information needed to respond to the global pandemic, this operational audit focused on COVID-19 data collection and reporting processes at the Agency for Health Care Administration (Agency), Department of Health (Department), and Division of Emergency Management (Division) during the period March 1, 2020, through October 9, 2020,” the report said. Now, the state’s Auditor General has found the case and death data for COVID-19 from FDOH was inaccurate, to some extents. The findings concerning Jones’ whistleblower allegations, that FDOH was directed to falsify positivity rates, that new case data on positivity was misrepresented, and that Jones was directed to restrict access to underlying data, were all reported false in the OIG report. The report from the FDOH OIG found the claims by Jones were “unsubstantiated” and “unfounded.” Avera Hand Co.COVID-19 may affect babies’ brain development in womb, study says.Avera De Smet Memorial Hospital - De Smet.Avera Dells Area Hospital - Dell Rapids.Check with your healthcare provider about the use of monoclonal antibodies or an antiviral. Treatment for COVID-19 is available in many parts of the state. Individuals who meet high-risk criteria and test positive should contact their primary care physician about a referral for antibody treatment within three days of a positive test result and no later than 10 days after symptom onset. Having a medical-related technological dependence (e.g, tracheostomy, gastrostomy, or positive pressure ventilation ).Neurodevelopmental disorders (e.g., cerebral palsy) or other complex conditions (e.g., genetic or metabolic syndromes and severe congenital abnormalities).Chronic lung diseases (e.g., chronic obstructive pulmonary disease, asthma, interstitial lung disease, cystic fibrosis, and pulmonary hypertension).Cardiovascular disease (including congenital heart disease) or hypertension.Having a body mass index (BMI) greater than 25 (overweight or obese).Currently receiving immunosuppressive treatment.Examples of chronic medical conditions include: Are at high risk of progressing to severe COVID-19 infection or of needing to be admitted to a hospital because of COVID-19.Are age 12 or older and weigh at least 88 pounds and.Are within 10 days of the start of their symptoms.Who can get this treatment? Antibody treatment can be used by people with mild to moderate COVID-19 who:.The antibodies bind to the spike protein of the COVID-19 virus to stop the virus from entering your cells and continuing the infection. Bamlanivimab + etesevimab and casirivimab + imdevimab are monoclonal antibodies that are specifically designed to protect against severe COVID-19 infection. What are monoclonal antibodies? Monoclonal antibodies are laboratory-made proteins that mimic the antibodies created by your immune system to fight off harmful viruses. View the South Dakota Monoclonal Antibody Treatment flyer Food and Drug Administration issued an Emergency Use Authorization to allow the use of monoclonal antibodies for the treatment of mild to moderate symptoms of COVID-19 in adults and pediatric patients. SD Childhood Lead Poisoning Prevention Program. 
Middle Eastern Respiratory Syndrome (MERS).  
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
